BD DISP SYR INS .5CC 30G BP
SKU: 110339BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD DISP SYR INS .5CC 30G SC (10’S)
SKU: 110340BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD DISP SYR INS .5CC 31G 6MM SC (10’S)
SKU: 110341BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10
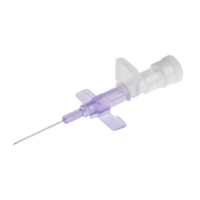
BD Insyte Autoguard
SKU: 110347, 110348, 110349, 110350Caring for you and your patients
✓ Provides safety and performance while enhancing the patient experience:
– No need to worry about stopping blood during initial insertion
– Smooth, tapered catheter tip which can help minimize insertion discomfort
✓ Push-button needle shielding along with blood control technology demonstrated:
– A statistically significant 95% reduction in needlesticks
– To reduce the risk of blood exposure by 95%
✓ Constructed with BD Vialon Biomaterial and BD Instaflash Needle Technology (20 to 24 gauge)
BD Insyte-W
SKU: 110352, 110353, 110354, 110355BD Vialon™, a unique proprietary biomaterial, has been proven to enhance clinical and economic outcomes.
- Increase the ease of penetration
- Reduces the risks of infiltration
- Exhibits greater kink resistance
- Reduces the risks of phlebitis up to 50%
- Reduces IV restarts due to reduced complications
- Increases indwelling time
- Reduces the total IV therapy costs to use due to longer indwelling time
Stabilizing wings provide a large surface area for taping down the catheter improving catheter securement.
Features:
- Product Type: Peripheral Venous Catheter
- DNR (Latex) free? Yes
- Sterile? Yes
- How sterilized: Ethylene Oxide
- Feature: DEHP free, Does not contain natural rubber latex
- Storage: Normal conditions
- Resterilized: No
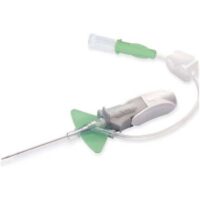
BD IV Cann Neoflon 24G
SKU: 110356 ✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle
BD IV Cann Neoflon 26G
SKU: 110357 ✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle
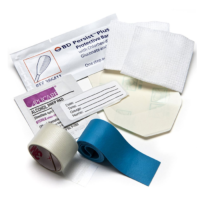
BD IV Start Pak W/ Tega 386170
SKU: 110359BD Persist™ skin prep provides one-step infection control with up to 5 days anti-microbial effectiveness. Tourniquet contains no rubber latex.
Accessories Include:
1 – Tourniquet (contains no natural rubber latex)
2 – 2 in. x 2 in. gauze sponge (5.1 cm x 5.1 cm)
1 – Roll Transpore™ tape (24 in x 3/4 in)
1 – Transparent Dressing
1 – Alcohol Swab
1 – BD Persist Povidone Iodine Prep
1 – Site Label
Product Features:
Product Type : IV Start Kit
DNR (Latex) Free: YES
Storage : Normal Conditions
Resterilize: NO
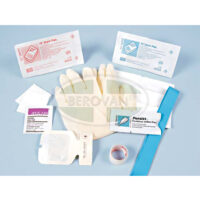
BD IV Start Pak with Persist with Gloves
SKU: 110358BD IV Start Pak™ with BD Persist™ skin prep, medium gloves, Tegaderm™ dressing (frame style), semipermeable dressing and accessories.
Accessories include:
- 2 – 2 in. x 2 in. gauze sponge (5.1cm x 5.1cm)
- 1 – Alcohol swab
- 1 – Tourniquet (contains no natural rubber latex)
- 1 – Site label
- 1 – Roll Transpore™ tape (24 in. x 3/4 in.)
- 1 – Transparent Dressing
Our high quality products offer a convenient sterile kit with everything needed to ensure optimal outcomes for all peripheral catheter placements. BD Persist contains 70% alcohol and 10% povidone iodine formulated in a unique polymer base, forming a barrier on the skin.
Feature:
BD Persist™ skin prep provides one-step infection control w/up to 5 days anti- microbial effectiveness, Glove & Tourniquet contains no natural rubber latex.
BD Nexiva 18GA 1.25IN HF Y
SKU: 110360✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.
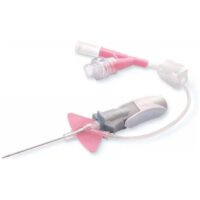
BD Nexiva 20GA 1.00IN HF Y
SKU: 110361✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.
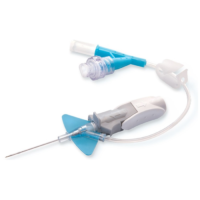
BD Nexiva 22GA 1.00IN Y 383532
SKU: 110362 ✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 24GA 0.56IN Y 383530
SKU: 110363✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.
BD Pen Needle
SKU: N/ABD Pen Needles are available in boxes of 100 and can be used as replacement needles for all insulin pens and dosers.
✓ At 5mm, the BD Ultra-Fine™ III Mini pen needle is the shortest pen needle available
✓ The BD Ultra-Fine™ III Mini pen needle requires no pinch-up while injecting
BD PEN NEEDLE 31G (5MM) 10’s
SKU: 110364BD Micro-Fine™ pen needles conveniently help deliver a precise dose, requiring minimal effort for people with diabetes—whether they are newly diagnosed or experienced insulin users. Designed for single-use and available on prescription in three different needle lengths, our pen needles support whatever needle length and injection technique patients need.
BD Micro-Fine pen needles support diabetic patients with the single-use design and needle length options.

BD PEN NEEDLE 32GX4 AP (4MM-PENTAPOINT) 10+1’S
SKU: 110739The BD Ultra-Fine™ 4mm pen needle is BD smallest, thinnest pen needle, promoting comfort. The pen needle incorporates EasyFlow™ technology, which improves insulin flow through the needle and makes the device easier to use, even for users with hand-strength challenges—enabling users to receive their full dose. The pen needle also features the patented PentaPoint™ Comfort five-bevel needle tip to promote a more comfortable and easier injection, compared to a three-bevel needle.
BD Ultra-FineTM 4mm Pen Needles combine TWO technologies to deliver Easier, Quicker & More Comfortable Injection Experience:
1. EasyflowTM technology
Extra Thin Wall needles for easier and quicker injection
2. PentapointTM technology
Five-bevel needle tip for more comfort.
BD PEN NEEDLE 32X4 AP (-PENTAPOINT)
SKU: 110368The BD Ultra-Fine™ 4mm pen needle is BD smallest, thinnest pen needle, promoting comfort. The pen needle incorporates EasyFlow™ technology, which improves insulin flow through the needle and makes the device easier to use, even for users with hand-strength challenges—enabling users to receive their full dose. The pen needle also features the patented PentaPoint™ Comfort five-bevel needle tip to promote a more comfortable and easier injection, compared to a three-bevel needle.
BD Ultra-FineTM 4mm Pen Needles combine TWO technologies to deliver Easier, Quicker & More Comfortable Injection Experience:
1. EasyflowTM technology
Extra Thin Wall needles for easier and quicker injection
2. PentapointTM technology
Five-bevel needle tip for more comfort.

BD DISP SYR INS .5CC 30G BP
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD DISP SYR INS .5CC 30G SC (10’S)
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD DISP SYR INS .5CC 31G 6MM SC (10’S)
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10
BD DISP SYR INS .5CC 31G SC(10’S)
SKU: 111827
BD Insyte Autoguard
Caring for you and your patients
✓ Provides safety and performance while enhancing the patient experience:
– No need to worry about stopping blood during initial insertion
– Smooth, tapered catheter tip which can help minimize insertion discomfort
✓ Push-button needle shielding along with blood control technology demonstrated:
– A statistically significant 95% reduction in needlesticks
– To reduce the risk of blood exposure by 95%
✓ Constructed with BD Vialon Biomaterial and BD Instaflash Needle Technology (20 to 24 gauge)

BD Insyte-W
BD Vialon™, a unique proprietary biomaterial, has been proven to enhance clinical and economic outcomes.
- Increase the ease of penetration
- Reduces the risks of infiltration
- Exhibits greater kink resistance
- Reduces the risks of phlebitis up to 50%
- Reduces IV restarts due to reduced complications
- Increases indwelling time
- Reduces the total IV therapy costs to use due to longer indwelling time
Stabilizing wings provide a large surface area for taping down the catheter improving catheter securement.
Features:
- Product Type: Peripheral Venous Catheter
- DNR (Latex) free? Yes
- Sterile? Yes
- How sterilized: Ethylene Oxide
- Feature: DEHP free, Does not contain natural rubber latex
- Storage: Normal conditions
- Resterilized: No

BD IV Cann Neoflon 24G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Cann Neoflon 26G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Start Pak W/ Tega 386170
BD Persist™ skin prep provides one-step infection control with up to 5 days anti-microbial effectiveness. Tourniquet contains no rubber latex.
Accessories Include:
1 – Tourniquet (contains no natural rubber latex)
2 – 2 in. x 2 in. gauze sponge (5.1 cm x 5.1 cm)
1 – Roll Transpore™ tape (24 in x 3/4 in)
1 – Transparent Dressing
1 – Alcohol Swab
1 – BD Persist Povidone Iodine Prep
1 – Site Label
Product Features:
Product Type : IV Start Kit
DNR (Latex) Free: YES
Storage : Normal Conditions
Resterilize: NO

BD IV Start Pak with Persist with Gloves
BD IV Start Pak™ with BD Persist™ skin prep, medium gloves, Tegaderm™ dressing (frame style), semipermeable dressing and accessories.
Accessories include:
- 2 – 2 in. x 2 in. gauze sponge (5.1cm x 5.1cm)
- 1 – Alcohol swab
- 1 – Tourniquet (contains no natural rubber latex)
- 1 – Site label
- 1 – Roll Transpore™ tape (24 in. x 3/4 in.)
- 1 – Transparent Dressing
Our high quality products offer a convenient sterile kit with everything needed to ensure optimal outcomes for all peripheral catheter placements. BD Persist contains 70% alcohol and 10% povidone iodine formulated in a unique polymer base, forming a barrier on the skin.
Feature:
BD Persist™ skin prep provides one-step infection control w/up to 5 days anti- microbial effectiveness, Glove & Tourniquet contains no natural rubber latex.

BD Nexiva 18GA 1.25IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 20GA 1.00IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 22GA 1.00IN Y 383532
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 24GA 0.56IN Y 383530
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Pen Needle
BD Pen Needles are available in boxes of 100 and can be used as replacement needles for all insulin pens and dosers.
✓ At 5mm, the BD Ultra-Fine™ III Mini pen needle is the shortest pen needle available
✓ The BD Ultra-Fine™ III Mini pen needle requires no pinch-up while injecting

BD PEN NEEDLE 31G (5MM) 10’s
BD Micro-Fine™ pen needles conveniently help deliver a precise dose, requiring minimal effort for people with diabetes—whether they are newly diagnosed or experienced insulin users. Designed for single-use and available on prescription in three different needle lengths, our pen needles support whatever needle length and injection technique patients need.
BD Micro-Fine pen needles support diabetic patients with the single-use design and needle length options.

BD PEN NEEDLE 32GX4 AP (4MM-PENTAPOINT) 10+1’S
The BD Ultra-Fine™ 4mm pen needle is BD smallest, thinnest pen needle, promoting comfort. The pen needle incorporates EasyFlow™ technology, which improves insulin flow through the needle and makes the device easier to use, even for users with hand-strength challenges—enabling users to receive their full dose. The pen needle also features the patented PentaPoint™ Comfort five-bevel needle tip to promote a more comfortable and easier injection, compared to a three-bevel needle.
BD Ultra-FineTM 4mm Pen Needles combine TWO technologies to deliver Easier, Quicker & More Comfortable Injection Experience:
1. EasyflowTM technology
Extra Thin Wall needles for easier and quicker injection
2. PentapointTM technology
Five-bevel needle tip for more comfort.

BD PEN NEEDLE 32X4 AP (-PENTAPOINT)
The BD Ultra-Fine™ 4mm pen needle is BD smallest, thinnest pen needle, promoting comfort. The pen needle incorporates EasyFlow™ technology, which improves insulin flow through the needle and makes the device easier to use, even for users with hand-strength challenges—enabling users to receive their full dose. The pen needle also features the patented PentaPoint™ Comfort five-bevel needle tip to promote a more comfortable and easier injection, compared to a three-bevel needle.
BD Ultra-FineTM 4mm Pen Needles combine TWO technologies to deliver Easier, Quicker & More Comfortable Injection Experience:
1. EasyflowTM technology
Extra Thin Wall needles for easier and quicker injection
2. PentapointTM technology
Five-bevel needle tip for more comfort.

BD DISP SYR INS .5CC 30G BP
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD DISP SYR INS .5CC 30G SC (10’S)
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD DISP SYR INS .5CC 31G 6MM SC (10’S)
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD Insyte Autoguard
Caring for you and your patients
✓ Provides safety and performance while enhancing the patient experience:
– No need to worry about stopping blood during initial insertion
– Smooth, tapered catheter tip which can help minimize insertion discomfort
✓ Push-button needle shielding along with blood control technology demonstrated:
– A statistically significant 95% reduction in needlesticks
– To reduce the risk of blood exposure by 95%
✓ Constructed with BD Vialon Biomaterial and BD Instaflash Needle Technology (20 to 24 gauge)

BD Insyte-W
BD Vialon™, a unique proprietary biomaterial, has been proven to enhance clinical and economic outcomes.
- Increase the ease of penetration
- Reduces the risks of infiltration
- Exhibits greater kink resistance
- Reduces the risks of phlebitis up to 50%
- Reduces IV restarts due to reduced complications
- Increases indwelling time
- Reduces the total IV therapy costs to use due to longer indwelling time
Stabilizing wings provide a large surface area for taping down the catheter improving catheter securement.
Features:
- Product Type: Peripheral Venous Catheter
- DNR (Latex) free? Yes
- Sterile? Yes
- How sterilized: Ethylene Oxide
- Feature: DEHP free, Does not contain natural rubber latex
- Storage: Normal conditions
- Resterilized: No

BD IV Cann Neoflon 24G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Cann Neoflon 26G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Start Pak W/ Tega 386170
BD Persist™ skin prep provides one-step infection control with up to 5 days anti-microbial effectiveness. Tourniquet contains no rubber latex.
Accessories Include:
1 – Tourniquet (contains no natural rubber latex)
2 – 2 in. x 2 in. gauze sponge (5.1 cm x 5.1 cm)
1 – Roll Transpore™ tape (24 in x 3/4 in)
1 – Transparent Dressing
1 – Alcohol Swab
1 – BD Persist Povidone Iodine Prep
1 – Site Label
Product Features:
Product Type : IV Start Kit
DNR (Latex) Free: YES
Storage : Normal Conditions
Resterilize: NO

BD IV Start Pak with Persist with Gloves
BD IV Start Pak™ with BD Persist™ skin prep, medium gloves, Tegaderm™ dressing (frame style), semipermeable dressing and accessories.
Accessories include:
- 2 – 2 in. x 2 in. gauze sponge (5.1cm x 5.1cm)
- 1 – Alcohol swab
- 1 – Tourniquet (contains no natural rubber latex)
- 1 – Site label
- 1 – Roll Transpore™ tape (24 in. x 3/4 in.)
- 1 – Transparent Dressing
Our high quality products offer a convenient sterile kit with everything needed to ensure optimal outcomes for all peripheral catheter placements. BD Persist contains 70% alcohol and 10% povidone iodine formulated in a unique polymer base, forming a barrier on the skin.
Feature:
BD Persist™ skin prep provides one-step infection control w/up to 5 days anti- microbial effectiveness, Glove & Tourniquet contains no natural rubber latex.

BD Nexiva 18GA 1.25IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 20GA 1.00IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 22GA 1.00IN Y 383532
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 24GA 0.56IN Y 383530
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Pen Needle
BD Pen Needles are available in boxes of 100 and can be used as replacement needles for all insulin pens and dosers.
✓ At 5mm, the BD Ultra-Fine™ III Mini pen needle is the shortest pen needle available
✓ The BD Ultra-Fine™ III Mini pen needle requires no pinch-up while injecting

BD PEN NEEDLE 31G (5MM) 10’s
BD Micro-Fine™ pen needles conveniently help deliver a precise dose, requiring minimal effort for people with diabetes—whether they are newly diagnosed or experienced insulin users. Designed for single-use and available on prescription in three different needle lengths, our pen needles support whatever needle length and injection technique patients need.
BD Micro-Fine pen needles support diabetic patients with the single-use design and needle length options.

BD PEN NEEDLE 32GX4 AP (4MM-PENTAPOINT) 10+1’S
The BD Ultra-Fine™ 4mm pen needle is BD smallest, thinnest pen needle, promoting comfort. The pen needle incorporates EasyFlow™ technology, which improves insulin flow through the needle and makes the device easier to use, even for users with hand-strength challenges—enabling users to receive their full dose. The pen needle also features the patented PentaPoint™ Comfort five-bevel needle tip to promote a more comfortable and easier injection, compared to a three-bevel needle.
BD Ultra-FineTM 4mm Pen Needles combine TWO technologies to deliver Easier, Quicker & More Comfortable Injection Experience:
1. EasyflowTM technology
Extra Thin Wall needles for easier and quicker injection
2. PentapointTM technology
Five-bevel needle tip for more comfort.

BD PEN NEEDLE 32X4 AP (-PENTAPOINT)
The BD Ultra-Fine™ 4mm pen needle is BD smallest, thinnest pen needle, promoting comfort. The pen needle incorporates EasyFlow™ technology, which improves insulin flow through the needle and makes the device easier to use, even for users with hand-strength challenges—enabling users to receive their full dose. The pen needle also features the patented PentaPoint™ Comfort five-bevel needle tip to promote a more comfortable and easier injection, compared to a three-bevel needle.
BD Ultra-FineTM 4mm Pen Needles combine TWO technologies to deliver Easier, Quicker & More Comfortable Injection Experience:
1. EasyflowTM technology
Extra Thin Wall needles for easier and quicker injection
2. PentapointTM technology
Five-bevel needle tip for more comfort.

BD DISP SYR INS .5CC 30G BP
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD DISP SYR INS .5CC 30G SC (10’S)
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD DISP SYR INS .5CC 31G 6MM SC (10’S)
BD Insulin Syringes with Ultra-Fine™ Needle
- Disposable single use insulin syringes with permanently – attached needle
- Polypropylene plastic suitable for medical use
- Latex free rubber stopper to avoid toxicity
- 30 degrees angle cut needle to reduce tissue trauma
- Electropolishes needle surface and micro-bonded lubrication to minimize pain
- No dead space design for higher accuracy and minimal wastage
- Large single scale markings and thin line plunger tip for accurate reading
- Available for different strengths of insulin, U40 and U100
- ETO sterilized with a sterility assurance level {SAL} of 10

BD Insyte Autoguard
Caring for you and your patients
✓ Provides safety and performance while enhancing the patient experience:
– No need to worry about stopping blood during initial insertion
– Smooth, tapered catheter tip which can help minimize insertion discomfort
✓ Push-button needle shielding along with blood control technology demonstrated:
– A statistically significant 95% reduction in needlesticks
– To reduce the risk of blood exposure by 95%
✓ Constructed with BD Vialon Biomaterial and BD Instaflash Needle Technology (20 to 24 gauge)

BD Insyte-W
BD Vialon™, a unique proprietary biomaterial, has been proven to enhance clinical and economic outcomes.
- Increase the ease of penetration
- Reduces the risks of infiltration
- Exhibits greater kink resistance
- Reduces the risks of phlebitis up to 50%
- Reduces IV restarts due to reduced complications
- Increases indwelling time
- Reduces the total IV therapy costs to use due to longer indwelling time
Stabilizing wings provide a large surface area for taping down the catheter improving catheter securement.
Features:
- Product Type: Peripheral Venous Catheter
- DNR (Latex) free? Yes
- Sterile? Yes
- How sterilized: Ethylene Oxide
- Feature: DEHP free, Does not contain natural rubber latex
- Storage: Normal conditions
- Resterilized: No

BD IV Cann Neoflon 24G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Cann Neoflon 26G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Start Pak W/ Tega 386170
BD Persist™ skin prep provides one-step infection control with up to 5 days anti-microbial effectiveness. Tourniquet contains no rubber latex.
Accessories Include:
1 – Tourniquet (contains no natural rubber latex)
2 – 2 in. x 2 in. gauze sponge (5.1 cm x 5.1 cm)
1 – Roll Transpore™ tape (24 in x 3/4 in)
1 – Transparent Dressing
1 – Alcohol Swab
1 – BD Persist Povidone Iodine Prep
1 – Site Label
Product Features:
Product Type : IV Start Kit
DNR (Latex) Free: YES
Storage : Normal Conditions
Resterilize: NO

BD IV Start Pak with Persist with Gloves
BD IV Start Pak™ with BD Persist™ skin prep, medium gloves, Tegaderm™ dressing (frame style), semipermeable dressing and accessories.
Accessories include:
- 2 – 2 in. x 2 in. gauze sponge (5.1cm x 5.1cm)
- 1 – Alcohol swab
- 1 – Tourniquet (contains no natural rubber latex)
- 1 – Site label
- 1 – Roll Transpore™ tape (24 in. x 3/4 in.)
- 1 – Transparent Dressing
Our high quality products offer a convenient sterile kit with everything needed to ensure optimal outcomes for all peripheral catheter placements. BD Persist contains 70% alcohol and 10% povidone iodine formulated in a unique polymer base, forming a barrier on the skin.
Feature:
BD Persist™ skin prep provides one-step infection control w/up to 5 days anti- microbial effectiveness, Glove & Tourniquet contains no natural rubber latex.

BD Nexiva 18GA 1.25IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 20GA 1.00IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 22GA 1.00IN Y 383532
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 24GA 0.56IN Y 383530
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Pen Needle
BD Pen Needles are available in boxes of 100 and can be used as replacement needles for all insulin pens and dosers.
✓ At 5mm, the BD Ultra-Fine™ III Mini pen needle is the shortest pen needle available
✓ The BD Ultra-Fine™ III Mini pen needle requires no pinch-up while injecting

BD PEN NEEDLE 31G (5MM) 10’s
BD Micro-Fine™ pen needles conveniently help deliver a precise dose, requiring minimal effort for people with diabetes—whether they are newly diagnosed or experienced insulin users. Designed for single-use and available on prescription in three different needle lengths, our pen needles support whatever needle length and injection technique patients need.
BD Micro-Fine pen needles support diabetic patients with the single-use design and needle length options.

BD PEN NEEDLE 32GX4 AP (4MM-PENTAPOINT) 10+1’S
The BD Ultra-Fine™ 4mm pen needle is BD smallest, thinnest pen needle, promoting comfort. The pen needle incorporates EasyFlow™ technology, which improves insulin flow through the needle and makes the device easier to use, even for users with hand-strength challenges—enabling users to receive their full dose. The pen needle also features the patented PentaPoint™ Comfort five-bevel needle tip to promote a more comfortable and easier injection, compared to a three-bevel needle.
BD Ultra-FineTM 4mm Pen Needles combine TWO technologies to deliver Easier, Quicker & More Comfortable Injection Experience:
1. EasyflowTM technology
Extra Thin Wall needles for easier and quicker injection
2. PentapointTM technology
Five-bevel needle tip for more comfort.

BD PEN NEEDLE 32X4 AP (-PENTAPOINT)
The BD Ultra-Fine™ 4mm pen needle is BD smallest, thinnest pen needle, promoting comfort. The pen needle incorporates EasyFlow™ technology, which improves insulin flow through the needle and makes the device easier to use, even for users with hand-strength challenges—enabling users to receive their full dose. The pen needle also features the patented PentaPoint™ Comfort five-bevel needle tip to promote a more comfortable and easier injection, compared to a three-bevel needle.
BD Ultra-FineTM 4mm Pen Needles combine TWO technologies to deliver Easier, Quicker & More Comfortable Injection Experience:
1. EasyflowTM technology
Extra Thin Wall needles for easier and quicker injection
2. PentapointTM technology
Five-bevel needle tip for more comfort.














