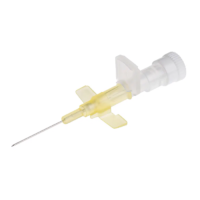
BD IV Cann Neoflon 24G
SKU: 110356 ✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle
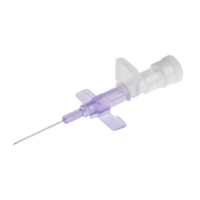
BD IV Cann Neoflon 26G
SKU: 110357 ✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle
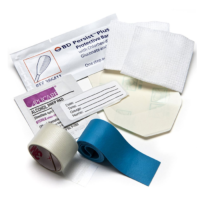
BD IV Start Pak W/ Tega 386170
SKU: 110359BD Persist™ skin prep provides one-step infection control with up to 5 days anti-microbial effectiveness. Tourniquet contains no rubber latex.
Accessories Include:
1 – Tourniquet (contains no natural rubber latex)
2 – 2 in. x 2 in. gauze sponge (5.1 cm x 5.1 cm)
1 – Roll Transpore™ tape (24 in x 3/4 in)
1 – Transparent Dressing
1 – Alcohol Swab
1 – BD Persist Povidone Iodine Prep
1 – Site Label
Product Features:
Product Type : IV Start Kit
DNR (Latex) Free: YES
Storage : Normal Conditions
Resterilize: NO
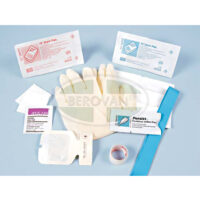
BD IV Start Pak with Persist with Gloves
SKU: 110358BD IV Start Pak™ with BD Persist™ skin prep, medium gloves, Tegaderm™ dressing (frame style), semipermeable dressing and accessories.
Accessories include:
- 2 – 2 in. x 2 in. gauze sponge (5.1cm x 5.1cm)
- 1 – Alcohol swab
- 1 – Tourniquet (contains no natural rubber latex)
- 1 – Site label
- 1 – Roll Transpore™ tape (24 in. x 3/4 in.)
- 1 – Transparent Dressing
Our high quality products offer a convenient sterile kit with everything needed to ensure optimal outcomes for all peripheral catheter placements. BD Persist contains 70% alcohol and 10% povidone iodine formulated in a unique polymer base, forming a barrier on the skin.
Feature:
BD Persist™ skin prep provides one-step infection control w/up to 5 days anti- microbial effectiveness, Glove & Tourniquet contains no natural rubber latex.
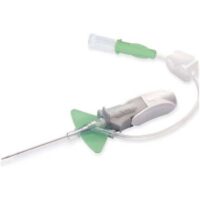
BD Nexiva 18GA 1.25IN HF Y
SKU: 110360✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.
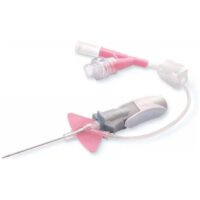
BD Nexiva 20GA 1.00IN HF Y
SKU: 110361✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD IV Cann Neoflon 24G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Cann Neoflon 26G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Start Pak W/ Tega 386170
BD Persist™ skin prep provides one-step infection control with up to 5 days anti-microbial effectiveness. Tourniquet contains no rubber latex.
Accessories Include:
1 – Tourniquet (contains no natural rubber latex)
2 – 2 in. x 2 in. gauze sponge (5.1 cm x 5.1 cm)
1 – Roll Transpore™ tape (24 in x 3/4 in)
1 – Transparent Dressing
1 – Alcohol Swab
1 – BD Persist Povidone Iodine Prep
1 – Site Label
Product Features:
Product Type : IV Start Kit
DNR (Latex) Free: YES
Storage : Normal Conditions
Resterilize: NO

BD IV Start Pak with Persist with Gloves
BD IV Start Pak™ with BD Persist™ skin prep, medium gloves, Tegaderm™ dressing (frame style), semipermeable dressing and accessories.
Accessories include:
- 2 – 2 in. x 2 in. gauze sponge (5.1cm x 5.1cm)
- 1 – Alcohol swab
- 1 – Tourniquet (contains no natural rubber latex)
- 1 – Site label
- 1 – Roll Transpore™ tape (24 in. x 3/4 in.)
- 1 – Transparent Dressing
Our high quality products offer a convenient sterile kit with everything needed to ensure optimal outcomes for all peripheral catheter placements. BD Persist contains 70% alcohol and 10% povidone iodine formulated in a unique polymer base, forming a barrier on the skin.
Feature:
BD Persist™ skin prep provides one-step infection control w/up to 5 days anti- microbial effectiveness, Glove & Tourniquet contains no natural rubber latex.

BD Nexiva 18GA 1.25IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 20GA 1.00IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD IV Cann Neoflon 24G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Cann Neoflon 26G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Start Pak W/ Tega 386170
BD Persist™ skin prep provides one-step infection control with up to 5 days anti-microbial effectiveness. Tourniquet contains no rubber latex.
Accessories Include:
1 – Tourniquet (contains no natural rubber latex)
2 – 2 in. x 2 in. gauze sponge (5.1 cm x 5.1 cm)
1 – Roll Transpore™ tape (24 in x 3/4 in)
1 – Transparent Dressing
1 – Alcohol Swab
1 – BD Persist Povidone Iodine Prep
1 – Site Label
Product Features:
Product Type : IV Start Kit
DNR (Latex) Free: YES
Storage : Normal Conditions
Resterilize: NO

BD IV Start Pak with Persist with Gloves
BD IV Start Pak™ with BD Persist™ skin prep, medium gloves, Tegaderm™ dressing (frame style), semipermeable dressing and accessories.
Accessories include:
- 2 – 2 in. x 2 in. gauze sponge (5.1cm x 5.1cm)
- 1 – Alcohol swab
- 1 – Tourniquet (contains no natural rubber latex)
- 1 – Site label
- 1 – Roll Transpore™ tape (24 in. x 3/4 in.)
- 1 – Transparent Dressing
Our high quality products offer a convenient sterile kit with everything needed to ensure optimal outcomes for all peripheral catheter placements. BD Persist contains 70% alcohol and 10% povidone iodine formulated in a unique polymer base, forming a barrier on the skin.
Feature:
BD Persist™ skin prep provides one-step infection control w/up to 5 days anti- microbial effectiveness, Glove & Tourniquet contains no natural rubber latex.

BD Nexiva 18GA 1.25IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 20GA 1.00IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD IV Cann Neoflon 24G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Cann Neoflon 26G
✓Suitable for patients with capillary brittleness, children and where a non-traumatic and delicate needle is requested.
✓ (Straight IV Cannula for paediatric / neonatal use)
✓ Butterfly Needle

BD IV Start Pak W/ Tega 386170
BD Persist™ skin prep provides one-step infection control with up to 5 days anti-microbial effectiveness. Tourniquet contains no rubber latex.
Accessories Include:
1 – Tourniquet (contains no natural rubber latex)
2 – 2 in. x 2 in. gauze sponge (5.1 cm x 5.1 cm)
1 – Roll Transpore™ tape (24 in x 3/4 in)
1 – Transparent Dressing
1 – Alcohol Swab
1 – BD Persist Povidone Iodine Prep
1 – Site Label
Product Features:
Product Type : IV Start Kit
DNR (Latex) Free: YES
Storage : Normal Conditions
Resterilize: NO

BD IV Start Pak with Persist with Gloves
BD IV Start Pak™ with BD Persist™ skin prep, medium gloves, Tegaderm™ dressing (frame style), semipermeable dressing and accessories.
Accessories include:
- 2 – 2 in. x 2 in. gauze sponge (5.1cm x 5.1cm)
- 1 – Alcohol swab
- 1 – Tourniquet (contains no natural rubber latex)
- 1 – Site label
- 1 – Roll Transpore™ tape (24 in. x 3/4 in.)
- 1 – Transparent Dressing
Our high quality products offer a convenient sterile kit with everything needed to ensure optimal outcomes for all peripheral catheter placements. BD Persist contains 70% alcohol and 10% povidone iodine formulated in a unique polymer base, forming a barrier on the skin.
Feature:
BD Persist™ skin prep provides one-step infection control w/up to 5 days anti- microbial effectiveness, Glove & Tourniquet contains no natural rubber latex.

BD Nexiva 18GA 1.25IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.

BD Nexiva 20GA 1.00IN HF Y
✓ Catheter’s stabilization platform minimizes movement that can lead to catheter complications, restarts and associated costs
– Built-in stabilization platform and pre-attached extension tubing
– Reduces dislodgement by 84%
– Meets the INS Standards and CDC Guidelines for catheters stabilization
✓ Proprietary BD Vialon™ Biomaterial enables longer dwell times, supporting the current infusion Nursing Standards of Practice for clinically indicated site rotation.
– In a 2014 clinical study, it was demonstrated that BD Nexiva™ catheters had a low rate of phlebitis and a median dwell time of 144 hours for catheters in place ≥ 24 hours
– In a 2014 clinical study, the longer dwell times of closed systems led to a cost reduction of approximately $1M/year/1,000 beds compared to a system
✓ Designed to minimize treatment delays and complications.
– Power injection compatible, minimizing the need to exchange catheter or tubing
– Single and dual ports and 18 – 22 gauge bore tubing are indicated for use in CT power injection
– Avoid leaking at the catheter/extension set connection due to the integrated system
✓ Integrated systems need fewer add-on devices, minimizing the number of manipulations which may lead to touch contamination and accidental disconnections
✓ BD Instaflash™ Needle Technology quick blood visualization may help improve insertion success, and therefore reduce insertion attempts
✓ BD Vialon™ Biomaterial has been clinically demonstrated to dwell up to 144 hours.














